 
He saw the electrons as fresh fruit in a positive-charged sphere, signifying the pudding. The atom, according to Thomson ’s concept, is structurally similar to plum pudding. OBSERVATION IN ACCORDANCE WITH THE THOMSON’S MODEL The plum pudding model was discarded both for simulation and practical grounds in favor of the Rutherford Atomic Theory, which describes electron orbits around a tiny positive nucleus. Although Thomson’s model could not accurately account for atomic structure, it served as a foundation for the creation of subsequent atomic models.Ītoms, according to Thomson, are homogeneous circles of positive electrode substances with electrons embedded inside. This model theoretically provided a detailed description of the atom’s interior structure. Many scientists proposed many theories and explanations for atom structure. Thomson asserted in 1897 that an atom’s fundamental body is spherical, including electrons (fine molecules inside the atom that form a repulsive force and an ionized “jelly” all around electrons that neutralize the charge. According to his idea, an atom contains millions of electrons, each of which is 2000 times smaller than a proton. He used a Cathode Ray Tube for his operation. Thomson’ had been an acclaimed physicist who is credited with finding ‘Electrons,’ after which he had been awarded the Nobel Prize. 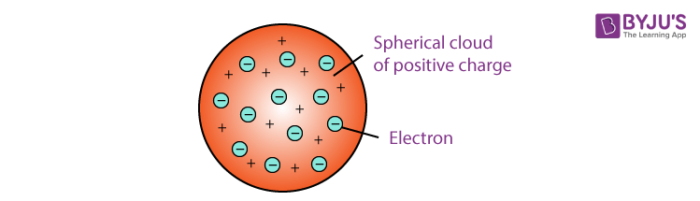
Thomson’s Atomic Model is a theoretical model that describes the interior atomic model. Sir Joseph Thomson, who also had discovered the electron earlier, was a staunch supporter. This concept theoretically provided the representation of an atom’s internal structure. In the year 1900, William Thomson proposed Thomson’s atomic model. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
